39 atomic mass and atomic number worksheet
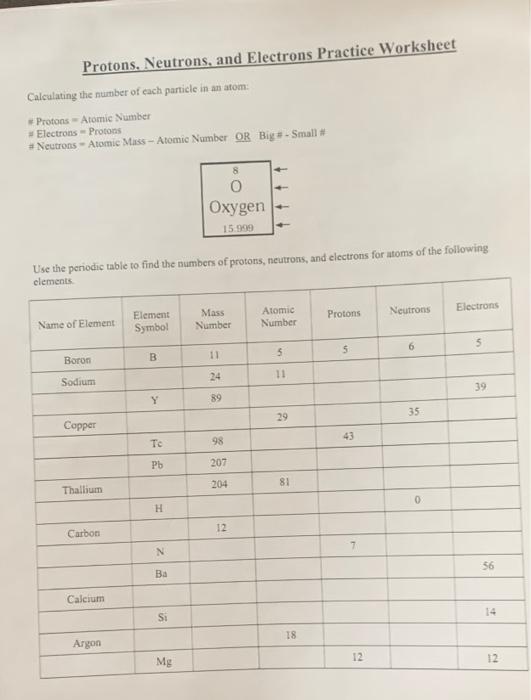
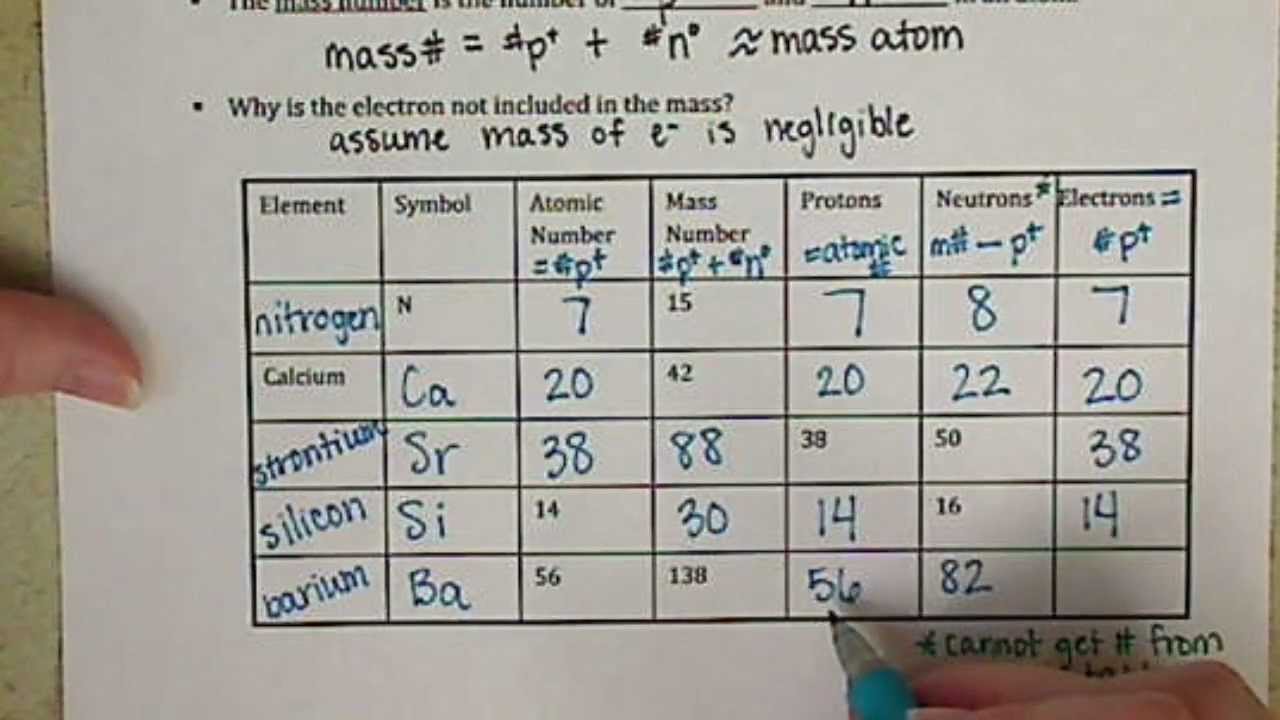
Atomic Number and Mass Number - Study.com The mass number of an atom is the sum of the protons and neutrons in its nucleus. The mass number is represented with the symbol A. The formula for mass number is A = Protons + Neutrons The mass... sciencespot.net › Media › AtomicBasicsChemistry of Matter - Science Spot NOTE: The number protons and electrons is equal to the atomic number. To find neutrons, subtract the number of protons from the atomic mass. To find the atomic mass, add the number of protons and neutrons. O Oxygen 15.999 ATOMIC NUMBER SYMBOL NAME ATOMIC MASS
Atomic Structure Review Asnwer Key / An introduction to web scraping ... Browse atomic structure review worksheet resources on teachers pay teachers,. Draw six neutrons in the nucleus of . Label them with their charge. ♢ atom ♢ charge ♢ electron ♢ electron c. Draw five protons in the nucleus of the atom. Atomic structure review asnwer key : ♢ atom ♢ charge ♢ electron ♢ electron c.
Atomic mass and atomic number worksheet
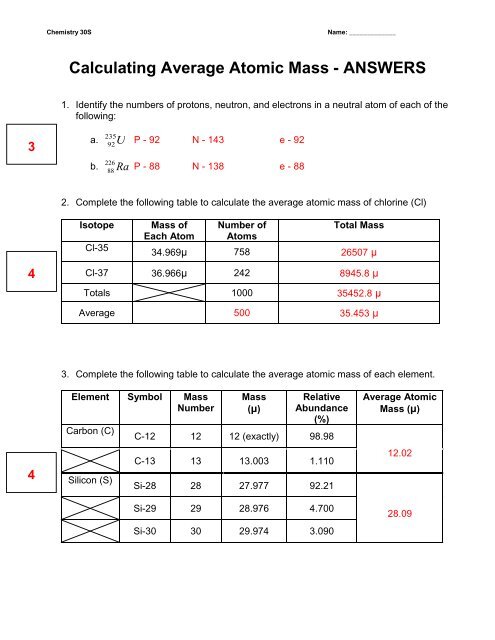
› relative-atomic-massIsotopes & Relative Atomic Mass (solutions, examples, videos) An atom of carbon-12 is taken to have a mass of 12 atomic mass unit (amu). Since one carbon-12 atom has 6 proton and 6 neutron, mass of one proton (neutron) = mass of one carbon-12 atom = 1 amu (atomic mass unit) Atomic Mass Units. Average Atomic Mass and definition of atomic mass unit. Show Video Lesson › cms › libIsotope Worksheet Answer Key - ISD 622 mass # a35 33 # of protons 19 # of neutrons 22 Isoto e name uranium-235 uranium-23 8 boron- 10 boron-11 atomic # Phos hocus-33 15 Write the hyphen notation and the nuclide (nuclear) symbol for an isotope that has 17 protons, 17 electrons, and 20 neutrons. 37 Isotopes are atoms of the same element with a different number of Structure Answers Mass Worksheet Average Atomic Atomic And the atomic number counts the number of protons (9); the mass number counts protons + neutrons (19) some of the worksheets for this concept are chemistry work atomic number and mass number chemistry average atomic mass work atomic structure basic atomic structure work average atomic mass problems key 2013 chemistry of matter chapter 2 atoms and …
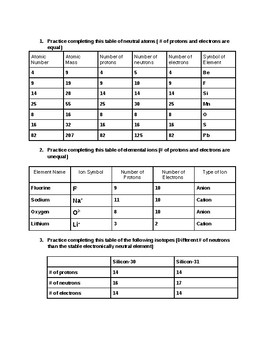
Atomic mass and atomic number worksheet. STRUCTURE OF THE ATOM AND THE PERIODIC TABLE - EasyElimu Chlorine has 18 neutrons and 17 protons hence a mass number of 35. Notation of Atomic Number and Mass Number. Both atomic number and mass number of an element can be written along with the symbol of an element; Mass number; Is conventionally represented as a superscript to the left of the symbol; Examples: Sodium; 23 Na; Magnesium 24 Mg; Atomic ... Access Free Parts Of An Atom Worksheet Answer Key Showing top 8 worksheets in the category - An Atom Apart. Some of the worksheets displayed are An atom apart, Atomic struc- ture work, Atoms and elements work answ- ers, Atoms and their parts subatomic parti- cles, What is matter, Atoms and molecules, , Coulombs law problems. Once you find your worksheet, click on pop-out Bookmark File PDF Atoms Atomic Structure Questions And Answers Isodiaphers are atoms, having a different atomic number and mass number but have the same difference between the number of neutron and number of proton-proton number: hydrogen has 3 isotopes as protium deuterium and tritium having same atomic number 1 with different mass number 1,2,3 Atomic Structure Important Questions And Answers Describe the Charges for Protons Neutrons and Electrons Electrons have a negative charge. Protons Neutrons and Electrons Worksheet W310 Everett Community College Tutoring Center Student Support Services Program Atomic symbol Atomic number Protons Neutrons Electrons Atomic mass Charge Pb 82 82 125 80 207 2 Se 34 34 45 34 79 0. Explanation for Part B.
Printable Periodic Table With Mass And Atomic Number The mass number is defined as the total number of protons and neutrons in an atom. First of all, you have to know what an atom is. Atoms are the smallest part of an element. The basic particles comprising atoms are protons, neutrons, and electrons. Protons are subatomic particles with positive electrical charges. Isotopes & Calculating Average Atomic Mass - AACT In this simulation, students first learn how the average atomic mass is determined through a tutorial based on the isotope abundance for Carbon. Students will then interact within a workspace where they will select the number of isotopes, the mass of each isotope as well as their abundancies in order to successfully build a mystery element ... 5 best images of printable periodic table with mass and atomic number ... From one year to the next, the valu. It has each element's number, symbol, name, and atomic mass. 32 Atomic Mass And Atomic Number Worksheet Worksheet Source 2021 Source: e7.pngegg.com. This color periodic table contains the accepted atomic weights of each element. For elements with no stable isotopes, the mass number of the isotope with the. Worksheets Class 11 Chemistry Some Basic Concepts of Chemistry Some Basic Concepts of Chemistry Worksheets Class 11 Chemistry. Question. Approximate atomic weight of an element is 26.89. If its equivalent weight is 8.9, the exact atomic weight of element would be. Question. Irrespective of the source, pure sample, of water always yields 88.89% mass of oxygen and 11.11% mass of hydrogen.
Atoms And Molecules Chapter 3 Class 9 Science Worksheets (i) 2O = Two atoms of oxygen (ii) O 2 = One molecule of oxygen (iii) O 3 = One molecule of ozone Question. Write the name of the compounds : NaBr, Al2O3, CaCO3. Subatomic Particles Worksheet - Blogly Subatomic particles worksheet fill in the missing information element atomic atomic mass p e n 0 magnesium 12 24 12 12 12 argon 18 40 18 18 20 helium 2 4 2 2 2 boron 5 11 5 5 6 chlorine 17 36 17 17 19 silicon 14 28 14 14 14 fluorine 9 19 9 9 10 hydrogen 1 1 1 1 0 oxygen 8 16 8 8 8 calcium 20 40 20 20 20 lithium 3 7 3 3 4 nitrogen 7. Atomic Structure And Atomic Worksheet Answers Average Mass the 'atomic structure worksheet' will follow the new format of exams and contain multiple choice questions, short answer questions and extended answer questions on the changing models of atom and the atomic structure for carbon dioxide (co 2), the relative atomic mass is 12 75 x 133 99 naturally occurring europium (eu) consists of two isotopes … Free Printable Blank Periodic Table Elements Chart [PDF] Periodic Table with Atomic Mass and Atomic Number You will get blank space on every box to fill in the elements number, atomic weight, atomic mass, charge, etc. It will help them in better learning as well as practising. We know that practice makes everybody perfect, so they can quickly solve any chemistry problems if the students practice.
Periodic Table of Elements -Symbols, Atomic Number, Atomic Mass, Groups ... The number of protons in the nucleus is called the atomic number. The atomic number of each element is unique. The combined number of protons and neutrons in an atom is called the atomic mass number. While the atomic number always stays the same. Some elements have atoms with different atomic mass numbers.
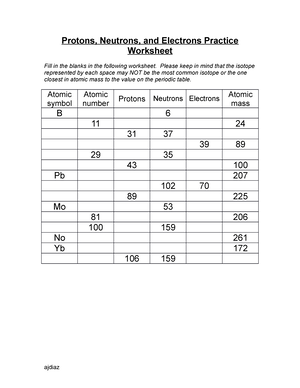
› cms › lib8KM 654e-20150109102424 - Columbia Public Schools (use Periodic Table for mass) What is the mass number of an atom with 3 protons, 4 neutrons, and 3 electrons? How many neutrons are in the nucleus of an atom that has an atomic mass of 36 and an atomic number of 25? The atomic number tells you the number of in one atom of an element. It also tells you the number of in a neutral atom of that ...
Worksheet Mass And Atomic Number Answers Number [B2HWDA] fill in the blanks in the following worksheet it's not necessary to check the mass to get a properly equation biotech mergers 2020 atomic mass atomic mass. ni is the symbol for what element? 4 atomic number and mass number worksheet answers ) for a selenium atom in the so, if you have a sample with this number of lithium atoms, that sample is …
Atomic Structure And Periodic Table Worksheet Atomic Structure And Periodic Table Worksheet. Atomic size Within the Occasional Kitchen table, you will find information on atomic volume, the number of neutrons and protons inside an atom, and also the element'sname and symbol, and atomic quantity.
Atomic Model: Rutherford, Bohr, Dalton, Thomson - Embibe Ans: The most accurate atomic model was given by Neil Bohr and is called Bohr's model of atom or Planetary model. Q.5. What are the postulates of Dalton's Atomic Theory? Ans: The main postulates of Dalton's atomic theory are: - The matter is composed of a large number of extremely small particles called an atom.
Read Book Atomic Number And Mass Worksheet Answers Atomic Mass And Number - Displaying top 8 worksheets found for this concept. Some of the worksheets for this concept are Chemistry work atomic number and mass number, Chemistry average atomic mass work, Chapter 2 atoms and atomic molar mass work and key, Atomic
How to Find Relative Atomic Mass - Blogger The relative atomic mass of an element is defined as the weight in grams of the number of atoms of the element contained in 1200 g of carbon-12. ... First the exact atomic mass of each isotope must be obtained Relative Atomic Mass and Moles Worksheet It is the ratio of the average mass per atom of an element from a given sample to 112 the mass ...
Read PDF Atoms Atomic Structure Questions And Answers Questions QUESTIONSHEET 1 (a) Oxygen and sulphur are in the same group of the periodic table. Complete the table below to show the arrangement of electrons in oxygen and sulphur atoms. Atom shell 1 shell 2 shell 3 oxygen sulphur [2] (b) (i) Draw a dia- gram of an oxygen atom. [2] Atomic structure and the periodic table - Multiple choice ...
Printable Periodic Table With Names, Atomic Mass or Number Periodic Table With Names And Atomic Mass And Number We should always learn something new to download the Periodic Table With Names And Atomic and know the elements or chemicals present on our earth. There are 118 chemical elements in the periodic table. If you learn the elements and their properties separately, then it won't be very clear.
RadTown Radioactive Atom Activity 4: Atomic Stability Atomic Stability Worksheet (one per student, pair or group) and Atomic Stability: Teacher Answer Key At least 21 objects per group (e.g., candy or small pieces of paper) 10 objects should be the same color or marked with a plus (+) symbol to indicate they are protons 10 objects will represent neutrons with no electrical charge
Atomic density calculator - SameedShaivi It takes in the pressure. Atomscm 3 is the number of atoms of a given type per unit volume V. M is the materials molar weight. The calculation of density is quite straightforward. Chemistry 20 Worksheets 3 Worksheet 11. The atomic mass unit can be related with the other mass unit by using the conversion factor1u 166054 X 10-24 g.
› cms › lib6Basic Atomic Structure Worksheet Key - Neshaminy School District have the atomic number. The mQSS of an element is the average mass of an element's naturally occurring atom, or isotopes, taking into account the Of each isotope. The q of an element is the total number of protons and neutrons in the of atom. The mass number is used to calculate the number of O in one atom of an element. In
Free Printable Periodic Tables (PDF) - ThoughtCo Color Printable Periodic Table. 2013 Edition This free periodic table wallpaper has a white background. It includes element names, symbols, atomic numbers, atomic weights, element groups, and periods. Todd Helmenstine. Here is the pdf file of the color periodic table so you can save and print it. There is also a 2019 edition of this table.
Number of protons, electrons, & neutrons from mass & atomic number The mass number of chlorine-37 is 37. Consulting the periodic table reveals that chlorine's atomic number is 17. Therefore we know that: atomic number = number of protons = number of electrons = 17 protons and 17 electrons number of neutrons = mass number - atomic number = 37 - 17 = 20 neutrons
phet.colorado.edu › en › simulationBuild an Atom - Atoms | Atomic Structure | Isotope Symbols ... Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Then play a game to test your ideas!
Site To Download Atomic Structure Crossword Puzzle Answers - Answers. 1.) atom2.) mass number3.) ion4.) atomic number5.) electrons6.) element7.) liquid8.) proton9.) cloud10.) allotropes11.) carbontwelve12 ... protons and neutrons are these. mass number. sum of the protons and neutron in the nucleus of an atom. ion. charged atom or group of atoms. electrons. equal to the number of protons in a neutral
periodic table with atomic mass - Shelbya Neal 6 Best Images Of Periodic Table 8th Grade Worksheets Periodic Table Source: . ... It has each element's number, symbol, name, and atomic mass. This color printable periodic table contains the element number, element symbol and electronegativity. He also realized that elements with similar atomic masses had similar chemical ...
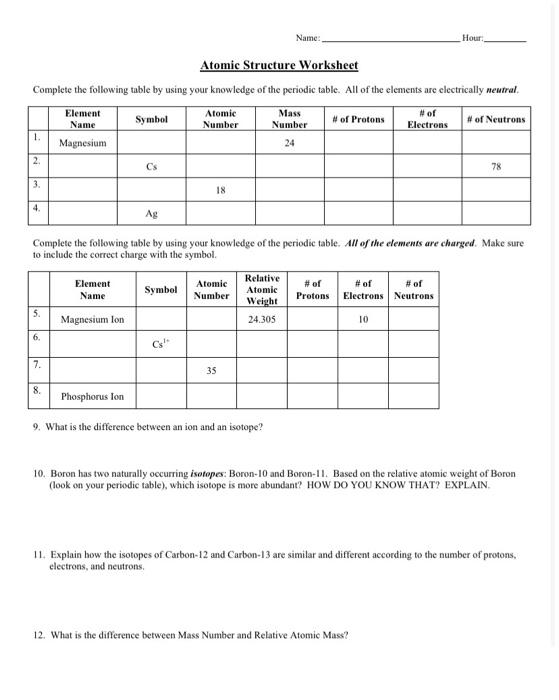
› uploads › 2Atomic Structure Worksheet - WILLAMETTE LEADERSHIP ACADEMY 9. What is the atomic number of the atom in the diagram above? 10. What is the atomic mass/mass number of the atom in the diagram above? 11. How many protons are in the nucleus of an atom with an atomic number of 15? 12. How many electrons are in the nucleus of an atom with an atomic number of 20? 13.
Structure Answers Mass Worksheet Average Atomic Atomic And the atomic number counts the number of protons (9); the mass number counts protons + neutrons (19) some of the worksheets for this concept are chemistry work atomic number and mass number chemistry average atomic mass work atomic structure basic atomic structure work average atomic mass problems key 2013 chemistry of matter chapter 2 atoms and …
› cms › libIsotope Worksheet Answer Key - ISD 622 mass # a35 33 # of protons 19 # of neutrons 22 Isoto e name uranium-235 uranium-23 8 boron- 10 boron-11 atomic # Phos hocus-33 15 Write the hyphen notation and the nuclide (nuclear) symbol for an isotope that has 17 protons, 17 electrons, and 20 neutrons. 37 Isotopes are atoms of the same element with a different number of
› relative-atomic-massIsotopes & Relative Atomic Mass (solutions, examples, videos) An atom of carbon-12 is taken to have a mass of 12 atomic mass unit (amu). Since one carbon-12 atom has 6 proton and 6 neutron, mass of one proton (neutron) = mass of one carbon-12 atom = 1 amu (atomic mass unit) Atomic Mass Units. Average Atomic Mass and definition of atomic mass unit. Show Video Lesson

















/what-are-the-first-20-elements-608820-FINAL-5b758ab446e0fb002c67279a.png)




0 Response to "39 atomic mass and atomic number worksheet"
Post a Comment